 
In Europe, the Committee for Medicinal Products for Human Use of the European Medicines Agency recently adopted a positive opinion recommending approval of KEYTRUDA in combination with chemotherapy for this patient population. Food and Drug Administration in November 2020 and was subsequently granted regular approval in July 2021. In the U.S., KEYTRUDA was granted accelerated approval by the U.S. These OS data follow prior analyses from KEYNOTE-355 that showed KEYTRUDA plus chemotherapy resulted in a statistically significant and clinically meaningful improvement in progression-free survival (PFS) compared with chemotherapy alone for the first-line treatment of patients with mTNBC whose tumors expressed PD-L1 (CPS ≥10) and supported approval of this regimen in the U.S. We remain committed to continuing to advance scientific understanding in TNBC.” “These OS data add to a strong body of evidence evaluating the use of KEYTRUDA plus chemotherapy for appropriate patients with TNBC. Vicki Goodman, vice president, clinical research, Merck Research Laboratories. “With these new data, KEYNOTE-355 has now met both primary endpoints, improving progression-free and overall survival for the approximately 40% of patients from this trial with metastatic TNBC whose tumors expressed PD-L1 (CPS ≥10),” said Dr. Treatment-related adverse events led to discontinuation in 18.3% of patients in the KEYTRUDA plus chemotherapy arm and 11.0% of patients in the chemotherapy arm. The incidence of treatment-related adverse events (TRAEs) was similar among patients in the two treatment groups, with Grade 3-5 TRAEs occurring in 68.1% of patients in the KEYTRUDA plus chemotherapy arm and 66.9% of patients in the chemotherapy arm. 
There was no statistically significant difference in OS between the treatment groups in the CPS ≥1 population due to statistical testing hierarchy, formal testing was not performed in the intention-to-treat (ITT) population. 
“I am very encouraged to see these new overall survival data for the KEYTRUDA combination, demonstrating a 27% relative reduction in the risk of death compared to chemotherapy alone in patients with mTNBC whose tumors expressed PD-L1 (CPS ≥10).” Hope Rugo, director, Breast Oncology and Clinical Trials Education, University of California San Francisco (UCSF) Helen Diller Family Comprehensive Cancer Center. “Metastatic TNBC has the worst survival prognosis among breast cancer subtypes, and there is an urgent need for treatment options that improve survival,” said Dr. These data were presented today in an oral presentation at the European Society for Medical Oncology (ESMO) Congress 2021 (Abstract #LBA16). Although the trial was not powered to compare efficacy between treatment groups by different chemotherapy regimens, the increase in OS was observed for KEYTRUDA plus chemotherapy across the three chemotherapy choices. There was an increase of 6.9 months in median OS with KEYTRUDA plus chemotherapy compared to chemotherapy alone (23.0 months vs. In this study, KEYTRUDA plus chemotherapy reduced the risk of death by 27% (HR=0.73 p=0.0093) in patients with mTNBC whose tumors expressed PD-L1 (Combined Positive Score ≥10), as compared to chemotherapy alone. KEYTRUDA is the first anti-PD-1 therapy in combination with chemotherapy to demonstrate a statistically significant and clinically meaningful improvement in OS for these patients. 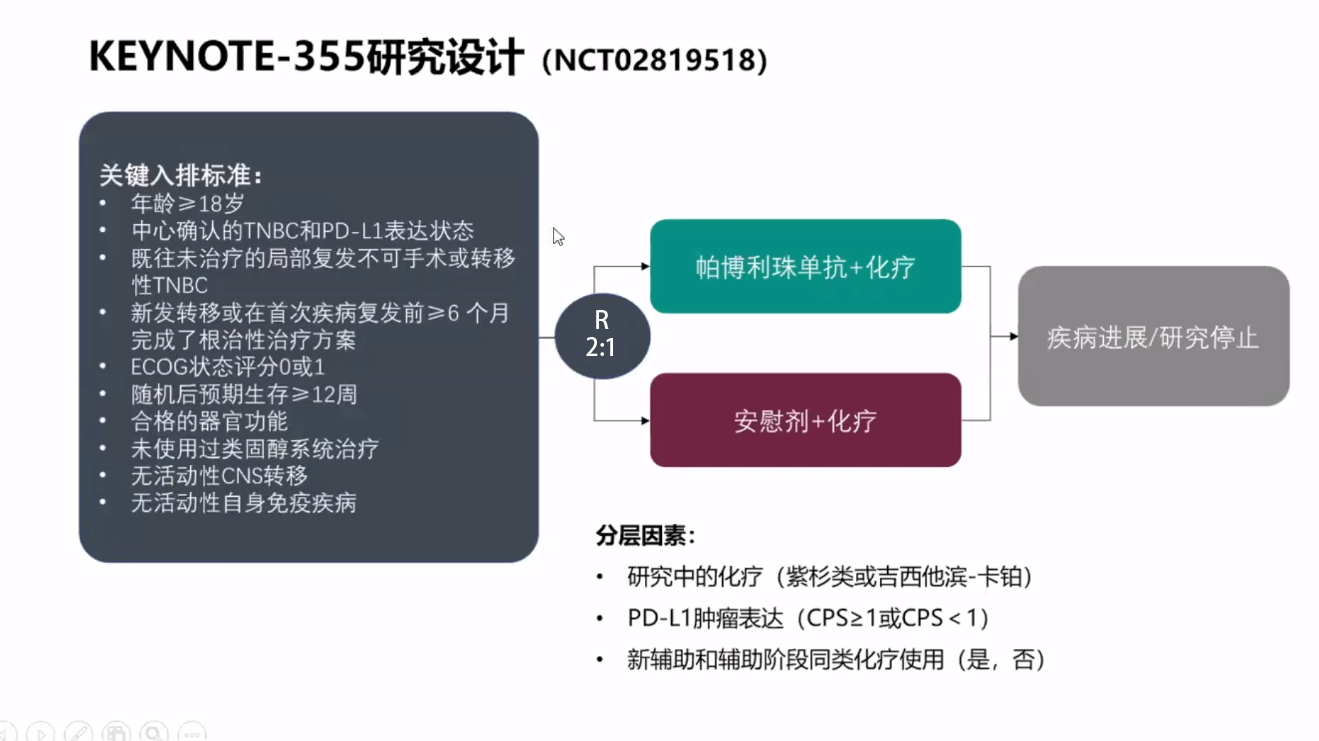
Merck (NYSE: MRK), known as MSD outside the United States and Canada, today announced the final overall survival (OS) results from the pivotal Phase 3 KEYNOTE-355 trial investigating KEYTRUDA, Merck’s anti-PD-1 therapy, in combination with chemotherapy (paclitaxel, nab-paclitaxel or gemcitabine/carboplatin) for the first-line treatment of patients with metastatic triple-negative breast cancer (mTNBC). First Anti-PD-1 Therapy in Combination With Chemotherapy to Demonstrate Statistically Significant Overall Survival for These Patientsĭata From Phase 3 KEYNOTE-355 Trial Presented at ESMO Congress 2021 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
